Keywords: biomarkers, TBI, S100B, GFAP, UCH-L1, TBI outcomes
What is the importance of biomarkers in acute TBI?
Biomarkers are defined by the National Institutes of Health Biomarkers Definitions Working Group as “a characteristic that is objectively measured and evaluated as an indicator of normal biological processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention.”[1,2].
Their role in the pathologic processes categorizez them into:
◉ diagnostic biomarkers
◉ monitoring biomarkers
◉ pharmacodynamic/response biomarkers
◉ predictive biomarkers
◉ prognostic biomarkers
◉ susceptibility or risk biomarkers, and safety biomarkers [2].
Beside the importance of biomarkers it is essential to know how can patients improve cognition after a TBI? and Why is TBI a global burden?
Biomarkers in Traumatic Brain Injury
Traumatic brain injury is defined as the dysfunction of the brain secondary to applying an external force. The various causes and mechanisms, clinical severity, and structural damage are part of the characteristics of TBI’s heterogeneity [3].
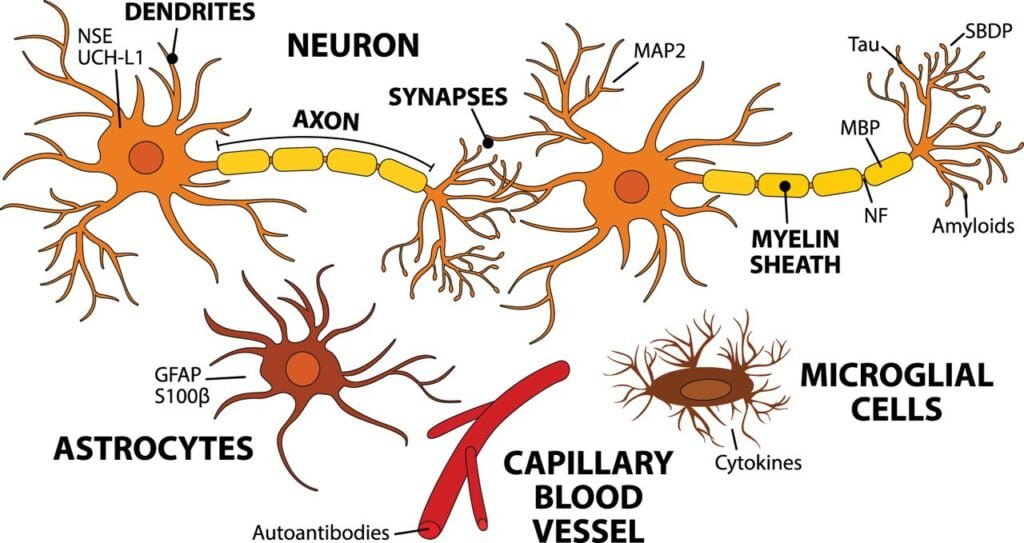
Fig. 1 – Biomarkers in acute TBI
from Harel, A. et al. “Biomarkers of traumatic brain injury: temporal changes in body fluids.” Eneuro 3.6 (2016)
The severity of TBI is of utter importance in the management, treatment, and prognosis of TBI patients [3,4]. It can be assessed clinically and with the help of imagery. While abnormal changes are usually visible on computer tomography (CT) and/or magnetic resonance imaging (MRI) in moderate and severe TBI cases, these may be overlooked in minor and mild TBI cases, particularly if the patient’s clinical presentation raises no red flags. There are certain advantages and disadvantages of CT scanning of the head, as presented in Figure 2.

Fig. 2 – Advantages & disadvantages of CT scanning
Radiation exposure represents one of the reasons why several guidelines have established different clinical criteria for the use of CT scans in TBI patients and one of the reasons for the careful selection of patients undergoing CT scans, particularly young ones.
MRI has the advantage of showing structural brain changes secondary to various lesions with higher sensitivity than CT. Furthermore, because MRI is nuclear magnetic resonance-based, radiation is not a problem. Unfortunately, some limitations of MRI reside in the higher cost, lower in-hospital availability, limited ability to show bone changes and recent bleeds compared to CT, and its common use in subacute and chronic settings.
The reasoning for using Biomarkes in TBI
The reasoning behind using biomarkers in TBI as a complementary test or even as a potential replacement test for imagery in the future is based on the anatomic-functional changes during TBI. If there is a blood-brain barrier dysfunction, it cannot be mirrored the types of imagery currently used in clinical settings, especially in mild TBI, mainly because of the reduced dimension of lesions (microscopic) [3,4]. TBI leads to several pathological changes according to the injury severity and type, including neuronal injury, axonal injury, astrogliosis, and cell death [5]. The role of the imagery is to assess the level of anatomical dysfunction secondary to TBI and to classify TBI in accordance with the severity [6].
The mechanisms of Biomarkes
With the help of contemporary TBI research, different pathogenetically heterogeneous mechanisms have been identified. Followingly, several biomarkers have been identified relating to the different mechanisms.
In case of a disrupted blood-brain barrier, including in TBI, different molecules will be able to enter and leave the central nervous system (CNS), thus, getting into the peripheral bloodstream. Different proteins usually found in the central nervous system reach the blood, hence consisting of an objective marker of the blood-brain-barrier disruption [3]. The biomarkers are usually present in important quantities in the CNS and cerebrospinal fluid (CSF) compared to the blood level. Furthermore, the quantity of the biomarkers rises in case of brain injury and can be assessed in blood and cerebrospinal fluid secondary to the blood-brain barrier dysfunction [3,4].
The research on biomarker levels in acute TBI has shown a correlation with injury severity, mirrored in the Glasgow Coma Scale, and with the presence of intracranial lesions on imagery [3,4].
Furthermore, obtaining biomarker levels peripherally represents an essential advantage in clinical settings [4]. That is why biomarkers represent an essential asset for detecting and assessing the severity of TBI.
Biomarker types
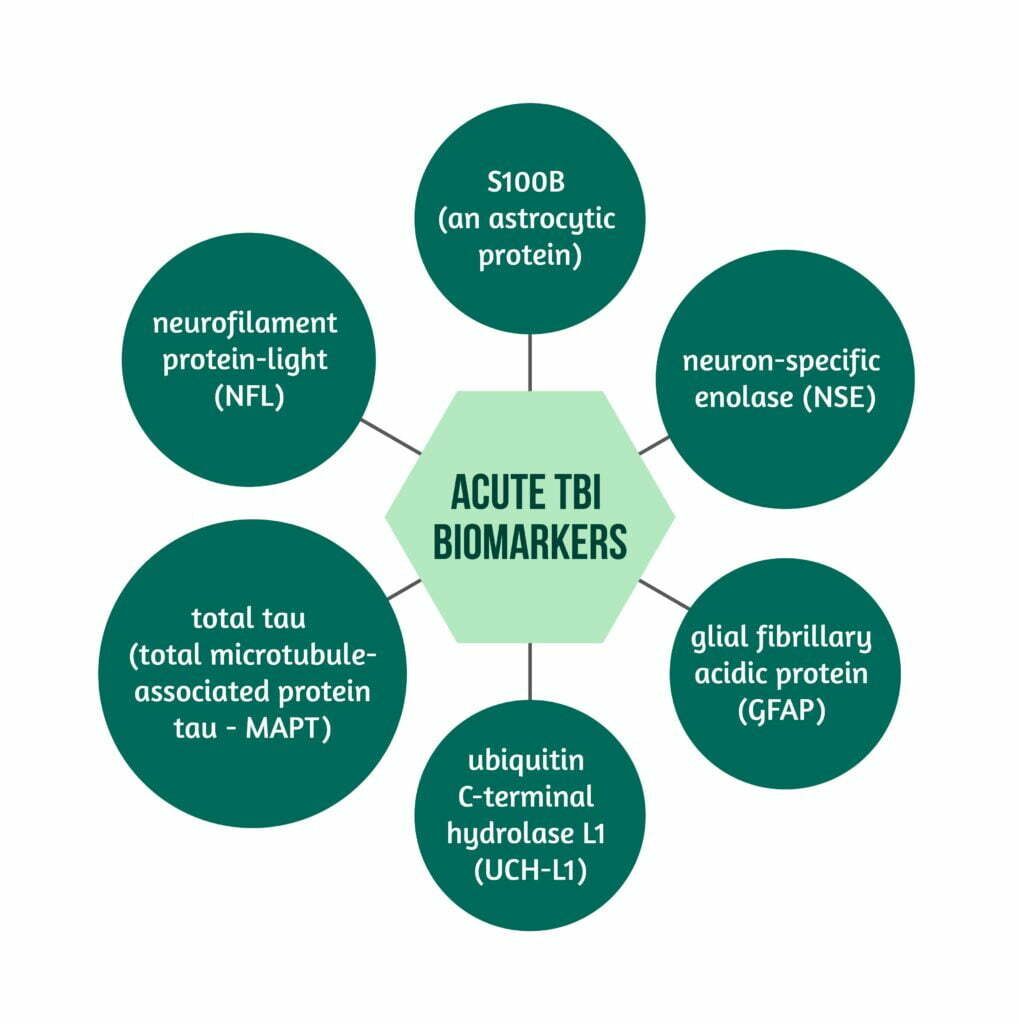
Several biomarkers are currently being researched for their role in acute TBI. Some of them are:
- S100B (an astrocytic protein)
- neuron-specific enolase (NSE)
- glial fibrillary acidic protein (GFAP)
- ubiquitin C-terminal hydrolase L1 (UCH-L1)
- neurofilament protein-light (NFL)
- total tau (total microtubule-associated protein tau – MAPT) [7]. These can be found in peripheral blood or CSF [4,5].
Where and when can biomarkers be used?
The easy access to biomarkers by examination of the biological fluids (CSF and blood) makes them a valuable tool in the emergency department, especially in cases of mild TBI when the imagery might not show significant lesions (despite the existence of microscopical injury). Thus, they can be used to evaluate the presence and severity of the injury. Furthermore, they could also be utilised as monitoring biomarkers, especially in intensive care units, where access to CSF and blood is easier and the sampling is carried out routinely. The athletes are a critical group of TBI patients that could benefit from these biomarkers. Similarly, the presence and severity of the TBI could be assessed and monitored for this group. Consequently, the decision-making of their return to practice would be assessed in all types of TBI, hopefully avoiding potential life-threatening injuries and posttraumatic sequelae. Moreover, another group that can benefit from the use of biomarkers is represented by military-related TBI patients.
The biomarkers as a predictive tool could be used for in-patient and out-patient settings.
Several biomarkers have been studied in regard to their role and involvement in TBI.
S100B
Currently, only S100B is integrated into clinical practice in some countries for its role in determining the necessity of CT scanning in patients with mild TBI, the marker having a high negative predictive value for the presence of intracranial lesions on head CT scan [7]. S100B is a marker for acute astroglial injury, and it can be detected in blood and CSF. It is the only biomarker currently being used daily in clinical practice because of its recommendation in the Scandinavian trauma guidelines.
S100B has been used as a TBI biomarker for some years, reducing the instances of unnecessary application of cranial CT, and increasing cost-effectiveness [8]. Although the levels of S100B were influenced by surgery or external ventricular drain, high levels in the serum were associated in all cases with mortality in TBI patients and subarachnoid hemorrhage. Furthermore, the use of imagery together with S100B improved the ability to predict clinical outcomes, particularly since another study showed the variation of S100B in accordance with the number and type of lesions in TBI patients [9].
NSE, UCH-1, and alpha-II spectrin
Revealing the presence of neuronal injury, biomarkers like NSE, UCH-1, and alpha-II spectrin have a significant potential to be used in clinical practice for TBI patients in the future. On the one hand, UCH-1 and alpha-II spectrin were elevated in TBI patients and correlated with the injury severity (GCS score) and mortality. On the other hand, the resulting parts of the alpha-II spectrin after breakdown have been identified and correlated with the outcome and severity of severe TBI patients [10]. Additionally, UCH-1 is known to point to neuronal cell body injury because it is situated in the neuronal body cytoplasm. Several studies have shown its increase in the blood and CSF of patients with mild TBI, severe TBI, people with sports concussions, and professional breacher trainees. Moreover, one study found the utility of this biomarker in the prognosis of TBI [9].
Furthermore, NSE proved to also be relatively elevated in hemolysis associated with cardiac arrest, thus leading, in some cases, to unspecificity [8,11]. Despite these findings, elevated NSE levels were found in patients with more severe TBI and consequent poorer outcomes. The administration of Memantine in patients with moderate TBI leads to a decrease in the NSE levels and an increase in GCS score. Last but not least, the correlation of high NSE levels was superior to that of S100B protein for mortality prediction after severe TBI [9].
GFAP
GFAP is an astroglial marker released secondary to TBI in the first posttraumatic hours, leading to elevated values in blood and CSF. The marker can be released as it is or as different fragments. Studies have shown that GFAP can point to different posttraumatic abnormalities of the brain, and its elevation can be correlated with the severity of the TBI and the patient’s outcome [11]. Furthermore, a study by Huebschmann et al. showed an association between GFAP levels in blood and CSF and the presence of brain changes on cranial CT. However, the level of GFAP in the biological liquids could not point out the TBI patients with poor functional outcome [12].
NFL, NPH, and tau protein
Other biomarkers that point to axonal injury are NFL (mainly spinal cord and profound brain regions), NPH, and tau (mainly cortical) proteins. Although all of the three biomarkers present high levels in patients with severe TBI, the NFL is considered the most important one due to its sensitivity being the highest.
TAU protein (microtubule-associated protein tau – MAPT)
Tau protein level was increased secondary to TBI, even without evidence of any macroscopic lesions on analyzed brains during autopsy. In addition, an increase in Tau protein levels in CSF has been observed in patients with severe TCE correlated with their outcome. For example, Bogoslovsky found the highest level seven days after the trauma [13]. Another study showed that increasing protein serum level was correlated with worse outcomes in patients with severe TBI [14].
Discussions
Many biomarkers have shown increased levels of acute TBI in serum and CSF [4,7].
However, research regarding the other biomarkers was scarce, thus making any clinical usage inadequate, although a study published in 2018 pointed to the possibility to use GFAP and UCH-L1 combined to differentiate between the presence and absence of lesions on CT in TBI patients [4,5]. Furthermore, another study from the US raised the possibility of using biomarkers to screen patients with no abnormal CT findings for the presence of abnormalities in brain MRI [4,5]. Nevertheless, some biomarkers, like GFAP and UCH-L1, have shown promising results in research conditions in this study. For example, GFAP was shown to have a high prediction rate of patients with lesions on CT scan, independent of injury severity, by its increase compared to the other biomarkers used independently and combined. Furthermore, research conditions in the study mentioned above have shown a correlation of the patient’s clinical status with mild TBI. In addition, GFAP and UCH-L1 used together have also shown a high prediction rate of the presence of CT lesions in TBI patients [7].
Over 500 biomarkers proved to be modified in patients with TBI, from different proteins to hormones, cytokines, genetic polymorphisms, coagulation markers, etc., thus providing information about the complexity of the brain damage secondary to TBI and potential use in clinical practice [15].
Consequently, serum and CSF biomarkers could represent an important asset for the diagnosis and prognosis through their correlation with the presence of brain lesions and injury severity on CT scans in acute TBI, serving simultaneously as an addon to clinical reasoning. Thus, they aid in the medical management of patients and represent a current research focus, aiming toward a possible future integration into different patient care algorithms. Furthermore, some TBI biomarkers are correlated with different outcome measures, like Glasgow Coma Scale (GOS), Glasgow Outcome Scale (GOS) and mortality rate [15].
Multiple biomarkers have a significant potential to aid physicians when treating patients with TBI in their clinical practice by improving the clinical decision-making process concerning injury severity and the presence of a lesion on CT scans. Different biomarkers have shown potential in this area. However, the different research methods used, the possibility of their usage in clinical settings, and the possibility of standardization and objectivization of tests need to be considered before any introduction into clinical practice. Currently, several studies are heterogeneous and not clinically applicable [4].
Several clinical trials currently analyze the utility of biomarkers, ranging from the use of biomarkers as secondary endpoints to therapeutic agents (for example, UCH-1 and GFAP) or to quantify blood biomarkers relating to the patient’s outcomes or presence of lesions on imagery.
Last but not least, further research must be done, as contemporary research regarding the use of biomarkers in acute TBI is insufficient.
References
- Biomarkers and surrogate endpoints: preferred definitions and conceptual framework. Biomarkers Definitions Working Group. Clin Pharmacol Ther 2001; 69(3):89-95. Retrieved from: https://doi.org/10.1067/mcp.2001.113989
- Jorge Manzanares, Francisco Sala, María Salud García Gutiérrez, Francisco Navarrete Rueda. Biomarkers,Reference Module in Biomedical Sciences. Elsevier 2021. Retrieved from: https://www.sciencedirect.com/science/article/pii/B9780128204726000608
- Zhang J, Puvenna V, Janigro D. Biomarkers of Traumatic Brain Injury and Their Relationship to Pathology. In: Laskowitz D, Grant G, editors. Translational Research in Traumatic Brain Injury. Boca Raton (FL): CRC Press/Taylor and Francis Group; 2016. Chapter 12. Retrieved from: https://www.ncbi.nlm.nih.gov/books/NBK326724/
- Czeiter E, Amrein K, Gravesteijn BY, et al. Blood biomarkers on admission in acute traumatic brain injury: Relations to severity, CT findings and care path in the CENTER-TBI study. EBioMedicine 2020;56:102785. DOI:10.1016/j.ebiom.2020.102785
- Shahim P, Politis A, van der Merwe A, Moore B, Ekanayake V, Lippa SM, Chou YY, Pham DL, Butman JA, Diaz-Arrastia R, Zetterberg H, Blennow K, Gill JM, Brody DL, Chan L. Time course and diagnostic utility of NFL, tau, GFAP, and UCH-L1 in subacute and chronic TBI. Neurology. 2020 Aug 11;95(6):e623-e636. DOI: 10.1212/WNL.0000000000009985.
- Thelin E, Al Nimer F, Frostell A, Zetterberg H, Blennow K, Nyström H, Svensson M, Bellander B-M, Piehl F, Nelson DW. Journal of Neurotrauma 2019.2850-2862.http://doi.org/10.1089/neu.2019.6375
- Harel Adrian, Kvist Mårten, Nuutinen Salla, Välimaa Lasse. eNeuro 2016, 3 (6) DOI: 10.1523/ENEURO.0294-16.2016
- Mastandrea P, Mengozzi S, Bernardini S. Systematic review and cumulative meta-analysis of the diagnostic accuracy of glial fibrillary acidic protein vs. S100 calcium-binding protein B as blood biomarkers in observational studies of patients with mild or moderate acute traumatic brain injury. Diagnosis (Berl) 2021. 1;9(1):18-27. DOI: 10.1515/dx-2021-0006.
- Mozaffari K, Dejam D, Duong C, et al. Systematic Review of Serum Biomarkers in Traumatic Brain Injury. Cureus 2021. 13(8):e17056. DOI: 10.7759/cureus.17056
- Wang KK, Yang Z, Zhu T, et al. An update on diagnostic and prognostic biomarkers for traumatic brain injury. Expert Rev Mol Diagn 2018. 18(2):165-180. DOI:10.1080/14737159.2018.1428089
- Chen S, Shi Q, Zheng S, et al. Role of α-II-spectrin breakdown products in the prediction of the severity and clinical outcome of acute traumatic brain injury. Exp Ther Med. 2016;11(5):2049-2053. DOI:10.3892/etm.2016.3153
- Huebschmann NA, Luoto TM, Karr JE, et al. Comparing Glial Fibrillary Acidic Protein (GFAP) in Serum and Plasma Following Mild Traumatic Brain Injury in Older Adults. Front Neurol 2020. 11:1054. DOI:10.3389/fneur.2020.01054
- Bogoslovsky T, Wilson D, Chen Y, et al. Increases of Plasma Levels of Glial Fibrillary Acidic Protein, Tau, and Amyloid β up to 90 Days after Traumatic Brain Injury. J Neurotrauma 2017;34(1):66-73. DOI:10.1089/neu.2015.4333
- Liliang PC, Liang CL, Weng HC, et al. Tau proteins in serum predict outcome after severe traumatic brain injury. J Surg Res 2010. 160(2):302-307. DOI:10.1016/j.jss.2008.12.022
- Edalatfar M, Piri SM, Mehrabinejad MM, et al. Biofluid Biomarkers in Traumatic Brain Injury: A Systematic Scoping Review. Neurocrit Care 2021. 35(2):559-572. DOI :10.1007/s12028-020-01173-1




